.
Similarly, it is asked, are all polar molecules covalent bonds?
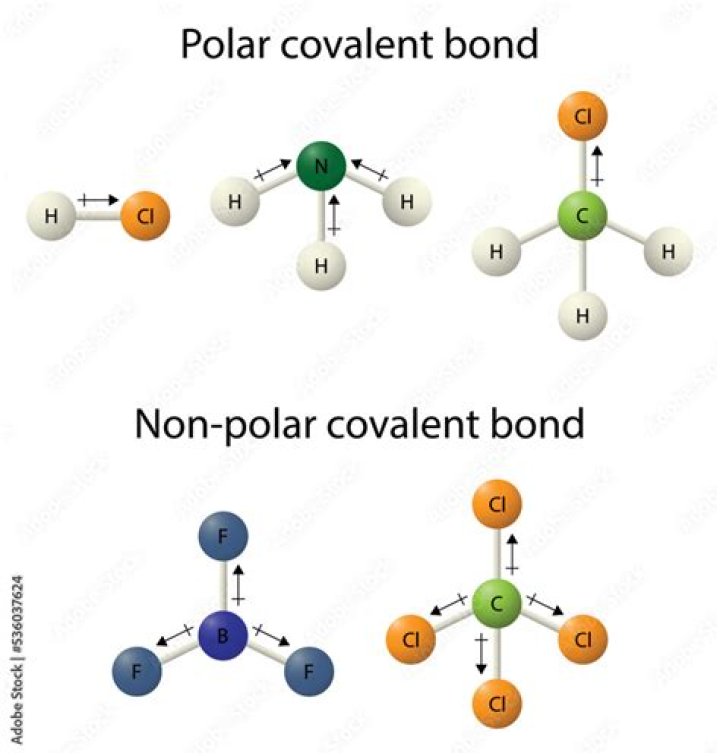
No. All covalent bonds between two different atoms are polar, only some are less polarised than others. On the other hand, covalent bonds between atoms of the same element are enerally considered non polar. However, this does not mean that polar molecules must have polar covalent bonds.
which molecule is a polar molecule? A polar molecule has a net dipole as a result of the opposing charges (i.e. having partial positive and partial negative charges) from polar bonds arranged asymmetrically. Water (H2O) is an example of a polar molecule since it has a slight positive charge on one side and a slight negative charge on the other.
Also, is a molecule polar or nonpolar?
Polar molecules occur when there is an electronegativity difference between the bonded atoms. Nonpolar molecules occur when electrons are shared equal between atoms of a diatomic molecule or when polar bonds in a larger molecule cancel each other out.
Is HCL polar or nonpolar?
HCL is a polar molecule as chlorine has a higher electronegativity than the hydrogen. Thus, it attracts electrons to spend more time at its end, giving it a negative charge and hydrogen a positive charge. How do you know if Br2 is polar or nonpolar?
Related Question AnswersIs CN a polar covalent bond?
Polar Molecules Both the C-H and the C-N bonds are polar. Nitrogen is more electronegative than carbon which is more electronegative than hydrogen. Each C-H bond is polar since carbon is more electronegative that hydrogen, and the C-Cl bond is polar since chlorine is more elctronegative than either carbon or hydrogen.Is n2 a polar covalent bond?
In the case of a diatomic, if the two atoms are different enough then it's considered polar. So, the molecules HF and HCl are polar because the halogen (F and Cl) attract the electrons much more than the H. In the molecule N2, both ends are the same. there is no difference so there is no polarity.Is NaCl a polar covalent bond?
Yes, NaCl is an ionic bond which makes it polar. A polar molecule consists of atoms having either a positive or negative bond. In this case, Na has a +1 charge and Cl has a -1 charge, making the bond polar. The difference in electronegativities is what makes a bond polar or nonpolar.What is a polar covalent molecule?
A polar bond is a covalent bond between two atoms where the electrons forming the bond are unequally distributed. This causes the molecule to have a slight electrical dipole moment where one end is slightly positive and the other is slightly negative.Is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.Is HCl ionic or covalent?
Sodium chloride is an ionic compound. Many bonds can be covalent in one situation and ionic in another. For instance, hydrogen chloride, HCl, is a gas in which the hydrogen and chlorine are covalently bound, but if HCl is bubbled into water, it ionizes completely to give the H+ and Cl- of a hydrochloric acid solution.Is water non polar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen. The electronegativity value of hydrogen is 2.1, while the electronegativity of oxygen is 3.5.Why is ClF polar?
The ClF bond is a polar covalent bond and the molecule has a net dipole moment (unlike CCl4 for instance where the bond dipoles cancel giving the molecule no net dipole moment). For these molecules, the applicable intermolecular interactions are dipole and induced dipole interactions.What are examples of polar and nonpolar molecules?
So there is a non zero net dipole moment and water is polar. So, the molecules with a non zero net dipole moment are called a polar molecules and if the net dipole moment is zero they are called nonpolar molecules.Examples of polar molecules include:
- water - H2O.
- ammonia - NH3.
- sulfur dioxide - SO2.