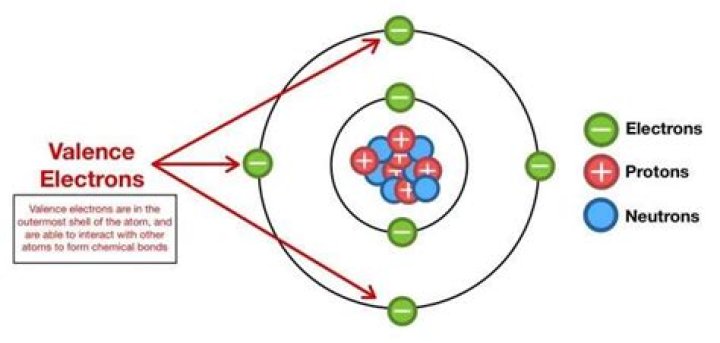
26 valence electrons
.
Moreover, how many valence electrons does phosphorus trichloride have?
5 valence electrons
Similarly, is pf3 trigonal planar? 10.32 Three electron groups: A trigonal planar molecular geometry has three bonding groups and no lone pairs of ~N. electrons so there are three electron pairs on atom A.
Also asked, how many valence electrons are in pf3?
26 valence electrons
Does phosphorus have a lone pair?
Phosphorus(V) fluoride, PF Phosphorus (in group 5) contributes 5 electrons, and the five fluorines 5 more, giving 10 electrons in 5 pairs around the central atom. Since the phosphorus is forming five bonds, there can't be any lone pairs.
Related Question Answers
What is the Lewis symbol for phosphorus?
There are a total of 40 valence electrons in the PCl5 Lewis structure. Remember when you draw the Lewis structure for PCl5 that Phosphorous (P) is in Period 3 on the Periodic table. This means that it can hold more than 8 valence electrons.Why is PCl3 trigonal pyramidal?
X3- three chlorine connected to the central phosphorus atom, N- total number of the lone pair of electrons. PCL3, lone pair of electrons is only one. And the actual shape of the Phosphorus Trichloride is Trigonal Pyramidal, and the bond angle of PCL3 is 109 degrees.Is PCl3 tetrahedral?
The shape of a P C l 3 PCl_3 PCl3 molecule is Trigonal pyramidal. The central P atom has one lone pair of electrons and three bond pairs of electrons. It undergoes s p 3 displaystyle sp^3 sp3 hybridisation which results in tetrahedral electron pair geometry and Trigonal pyramidal molecular geometry.What does the octet rule state?
The octet rule refers to the tendency of atoms to prefer to have eight electrons in the valence shell. When atoms have fewer than eight electrons, they tend to react and form more stable compounds.Is phosphorus trichloride a covalent or ionic bond?
Answer and Explanation: Phosphorus trichloride is a covalent molecule. A hint is the particular naming of the compound as it follows the rules for naming nonmetal covalentHow do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.Is phosphorus trichloride polar or nonpolar?
PCl3 is polar, because of its trigonal pyramidal structure. Only 3 of the 5 valence electrons of the phosphorus are used for bonding with chlorine, so the other two are unshared.How many valence electrons does c2h4 have?
12
How many lone pairs are in pf3?
one lone pair
What is the ideal angle between the phosphorus fluorine bonds?
PF5 Phosphorus Pentafluoride The resulting shape is a trigonal bipyramidal in which three fluorine atoms occupy equatorial and two occupy axial positions. The F-P-F angle between equatorial positions is 120°, between the axial and equatorial positions it is 90°.What is sbr2?
Standard Business Reporting (SBR2) The ATO accepts data for various reporting by business called Standard Business Reporting. The latest version of this electronic messaging is SBR2. It includes the following documents: Business Activity Statements (BAS)What is the Lewis structure for cs2?
There are 16 valence electrons for the CS2 Lewis structure. Carbon is the least electronegative atom and goes in the center of this structure. The Lewis structure for CS2 requires you have double bonds between the Carbon (C) and Sulfur atoms in order to fill the octet of Carbon.Why is pf3 a Lewis base?
PF3 on the other hand acts as a Lewis acid because the three fluorine atoms attached to it have highly electronegativity(4). And are electron deficient and hence pull the electron cloud towards themself,making the central atoms electron deficient.What is the Lewis structure for HF?
Drawing the Lewis Structure for HF Hydrogen has 1 valence electron and Fluorine (in Group 7 with F and Cl) has 7 valence electrons. With the Lewis Structure for HF remember that Hydrogen only needs 2 valence electrons to have a full outer shell. Be sure that you don't use more than the 8 valence electrons available.What is the Lewis structure for h2s?
Let's do the Lewis structure for H2S: Dihydrogen Sulfide. On the periodic table: Hydrogen, group 1, has 1 valence electron, but we have two Hydrogens here so let's multiply that by 2. Plus Sulfur is in group 6 or 16 on the periodic table, so it has 6 valence electrons. Total of 8 valence electrons.What is the shape of co2?
linear
Is bf3 planar?
Molecular Geometry of BF3 The geometry of molecule of BF3 is 'Trigonal Planar. ' With the reference of Chemistry, 'Trigonal Planar' is a model with three atoms around one atom in the middle.Is bcl3 polar or nonpolar?
The molecular geometry of BCl3 is trigonal planar with symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.Is h2o tetrahedral?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.