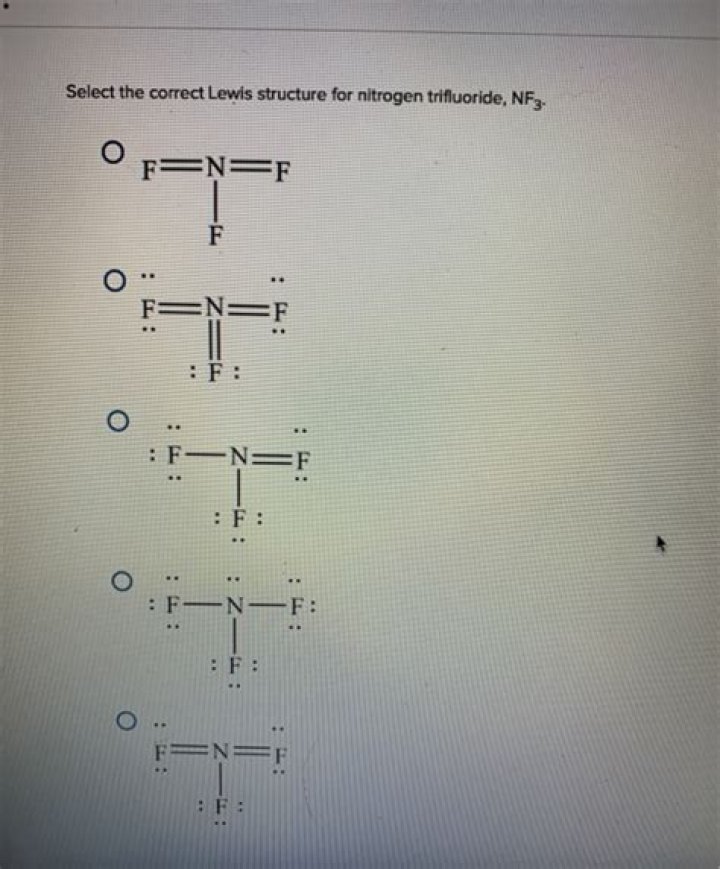
NF3 has a lone pair of electrons on the central nitrogen atom, giving it a trigonal pyramidal molecular geometry alongside the three N - F bonds. It is not able to accept electron pairs from other species and therefore is not a Lewis acid..
Beside this, what is the Lewis structure for nf3?
NF3 (Nitrogen trifluoride) is very similar to the NCl3 and NH3 Lewis structure. In the NF3 Lewis structure (and all structures) hydrogen goes on the outside. Hydrogen only needs two valence electrons to have a full outer shell. In the Lewis structure for NF3 there are a total of 8 valence electrons.
Likewise, which is strongest Lewis base? Nitrogen, being smallest in size, can give up its lone pair of electrons most easily. Hence, the N H 3 NH_{3} NH3 is the strongest Lewis base.
Subsequently, question is, why nh3 is stronger Lewis base than nf3?
So the 3 F atoms in NF3 attracts the lone pair of electrons in N and hence it becomes difficult for N to loose that pair of electrons, and therefore acts as a weak base. But in NH3, Hydrogen is less electronegative then Fluorine, and has less tendency to attract shared pair of electrons.
Why is nf3 least basic?
NF3 is least basic because F is most electronegative and it will attarct the lone pair of N atom and thus the electrons will the less available form donation.
Related Question Answers
What is the best Lewis structure for cs2?
Let's do the Lewis structure for CS2. On the periodic table, Carbon's in group 4, sometimes called 14, so it has 4 valence electrons. Sulfur in group 6 or 16, it has 6.How do you find the formal charge?
Formal Charge = [# valence electrons on neutral atom] – [(# lone electron pairs) + (½ # bonding electrons)] Valence electrons = corresponds to the group number of the periodic table (for representative elements). Lone Pairs = lone electrons sitting on the atom. Each electron counts as one and so a pair counts as two.How many lone pairs are in nf3?
In NF3 there are also three bond pairs, but the nitrogen has a lone pair as well. The four pairs of electrons arrange themselves tetrahedrally, but the description of the shape only takes account of the atoms. NF3 is pyramidal.Is nf3 tetrahedral?
NF3 is Trigonal Pyramidal, not Tetrahedral. There is no 4th molecule bonding with the nitrogen to make it Tetrahedral.What is h2cs?
Yes H2CS is a Polar Molecule. If you draw the Lewis Structure you have the central atom of Carbon single bonding to two hydrogen and double bonding to sulfur. There will be two lone pairs on Sulfur. This will satisfy the octet rule, and create this molecule.Does nf3 follow the octet rule?
An atom like phosphorus or sulfur which has more than an octet is said to have expanded its valence shell. This can only occur when the valence shell has enough orbitals to accommodate the extra electrons. Thus nitrogen can form NF3 (in which nitrogen has an octet) but not NF5.Which is the weakest Lewis base?
Lewis base is a compound that should donate electrons easily. The weakest Lewis base is C l ⊝ Cl^{circleddash} Cl⊝, because it has octet rule.Is alcl3 a Lewis acid?
With one empty orbital and three electron-withdrawing Cl atoms attached, the compound is thus an electron-acceptor at the Al center. By definition, that is a Lewis Acid.What makes a good Lewis base?
Lewis Bases are Nucleophilic meaning that they “attack” a positive charge with their lone pair. They utilize the highest occupied molecular orbital or HOMO (Figure 2). An atom, ion, or molecule with a lone-pair of electrons can thus be a Lewis base.Why does nh3 act as a Lewis base?
The proton is an electron -pair acceptor ,so it is a lewis acid. NH3 which has a lone pair of electron is an electron pair donor and therefore a lewis base. Ammonia is a weak base because its nitrogen atom has an electron pair that readily accepts a proton.Why is ammonia more basic than ph3?
es NH3 is more basic than PH3 as NH3 has more density of electron. Owing it to the small size of nitrogen and small P orbitals it has the ability to form stronger bonds due to effective overlap. In PH3 however, the large size of P results in the P orbitals becoming diffused preventing effective overlap.Which of the following is the strongest Lewis acid?
Boron tribromide is the STRONGEST Lewis acid known…and this is the experimental fact despite the reduced electronegativity of bromine as compared to fluorine, and chlorine.Why is BCl3 more acidic than bf3?
BCl3 is the stronger Lewis acid because the Boron center in BF3 participates in 2p(pi)-2p(pi) back bonding with the Fluorine atoms with a greater overlap thereby reducing it's deficiency.Which is more electronegative nh3 or nf3?
In NH3, N is the most electronegative element while in NF3, F is the most electronegative element. But in both the molecules, a lone pair of electrons is present. So,in addition to bond polarity and structure, the unshared pair of electrons also influence dipole moment values.What is Lewis base with example?
A Lewis base, then, is any species that has a filled orbital containing an electron pair which is not involved in bonding but may form a dative bond with a Lewis acid to form a Lewis adduct. For example, NH3 is a Lewis base, because it can donate its lone pair of electrons.What is Lewis acid with example?
Lewis Acids Examples include copper (Cu2), iron (Fe2+ and Fe3+), and hydrogen ion (H+). An atom, ion, or molecule with an incomplete octet of electrons can accept electrons.Is Iodine a Lewis acid or base?
Iodine is not an acid. It is a Halogen. Speaking of Halogens, you may know that the highly reactive and toxic Fluorine, Chlorine and Bromine are Halogens.Is BCl3 a Lewis acid?
The central atom in BCl3 (that is Boron, in trivalent state) has only six electrons around it — it is electron deficient and accepts electrons to complete it's octet. Therefore it acts as a Lewis acid by accepting electrons.How do you identify a Lewis base?
You draw the Lewis structure and look for a pair of electrons that can form a bond to another atom. A Lewis base is a molecule or ion that can donate a pair of electrons to form a bond. An atom, ion, or molecule with a lone-pair of electrons can be a Lewis base.