.
Besides, is the mobile or stationary phase more polar?
The longer the mobile phase travels, the better the separation between A and B. Stationary phases are usually very polar, while mobile phases vary widely in polarity, but are less polar than the stationary phase. This is called normal phase (NP) chromatography.
Beside above, how does stationary phase affect chromatography? Higher the adsorption to the stationary phase, the slower the molecule will move through the column. Higher the solubility in the mobile phase, the faster the molecule will move through the column.
In respect to this, what is the stationary and mobile phase in paper chromatography?
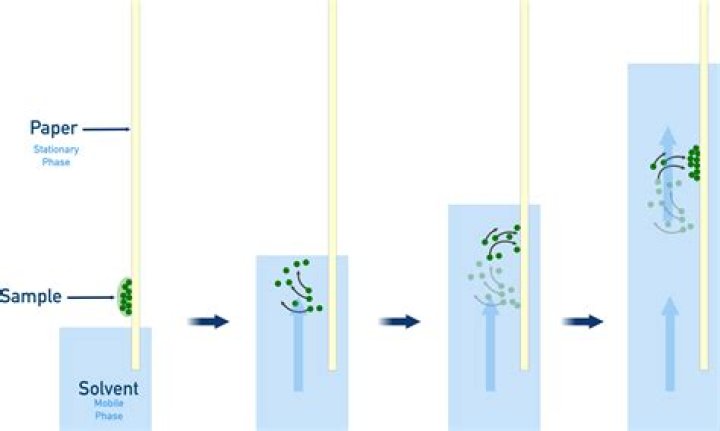
In paper chromatography, substances are distributed between a stationary phase and a mobile phase. The stationary phase is the water trapped between the cellulose fibers of the paper. The mobile phase is a developing solution that travels up the stationary phase, carrying the samples with it.
What is the stationary and mobile phase in TLC?
The silica gel (or the alumina) is the stationary phase. The stationary phase for thin layer chromatography also often contains a substance which fluoresces in UV light - for reasons you will see later. The mobile phase is a suitable liquid solvent or mixture of solvents.
Related Question AnswersIs silica polar or nonpolar?
silica gel is very polar. so more polar material moves more slowly than nonpolar material, which feels less attraction from the silica gel. it's used in TLC and column chromatography (not paper chromatography).What is the basic principle of paper chromatography?
Principle of paper chromatography: The principle involved is partition chromatography wherein the substances are distributed or partitioned between liquid phases. One phase is the water, which is held in the pores of the filter paper used; and other is the mobile phase which moves over the paper.What is Rf value?
The Rf value is defined as the ratio of the distance moved by the solute (i.e. the dye or pigment under test) and the distance moved by the the solvent (known as the Solvent front) along the paper, where both distances are measured from the common Origin or Application Baseline, that is the point where the sample isWhat do u mean by polarity?
Polarity refers to the physical properties of compounds such as boiling point, melting points and their solubilities. Definition of Polarity– “A state or a condition of an atom or a molecule having positive and also negative charges, especially in case of magnetic or an electrical poles.”What is polar mobile phase?
In normal-phase chromatography, the stationary phase is polar and the mobile phase is nonpolar. The mobile phase is generally a binary mixture of water and a miscible polar organic solvent like methanol, acetonitrile or THF. Retention increases as the amount of the polar solvent (water) in the mobile phase increases.Why is mobile phase nonpolar?
The mobile phase consists of a nonpolar solvent such as hexane or heptane mixed with a slightly more polar solvent such as isopropanol, ethyl acetate or chloroform. Retention decreases as the amount of polar solvent in the mobile phase increases.What property does TLC take advantage of?
TLC is an analytical tool widely used because of its simplicity, relative low cost, high sensitivity, and speed of separation. TLC functions on the same principle as all chromatography: a compound will have different affinities for the mobile and stationary phases, and this affects the speed at which it migrates.Why is stationary phase polar?
In normal phase chromatography, where the stationary phase is polar, polar molecules will spend more time adsorbed on the stationary phase, while less polar ones will be carried more quickly by the non-polar mobile phase.Why is water the stationary phase in paper chromatography?
This basically also goes for other kinds of thin layer chromatography, of which paper chromatography is only one technique. The idea is that the relatively polar molecules in the solid support material (e.g. Cellulose, silica, alumina etc.) adsorb water molecules so well that they will remain stationary.What is the purpose of water in paper chromatography?
Answer and Explanation: Water acts as a polar mobile phase in chromatography. It is more effective when used with a non polar stationary phase because there is a greaterWhat is the stationary phase in paper chromatography experiment?
The stationary phase in paper chromatography is the strip or piece of paper that is placed in the solvent. In thin-layer chromatography the stationary phase is the thin-layer cell. Both these kinds of chromatography use capillary action to move the solvent through the stationary phase.What does a high RF value mean in paper chromatography?
Rf = distance traveled by substance/distance traveled by solvent front. A high Rf (Ie 0.92) would refer to a substance that is very non-polar. Ie that substance moved a 92% of the entire distance the solvent traveled. A low Rf value (0.10) would refer to a substance that is very polar.What factors affect paper chromatography?
Retention factor values in thin layer chromatography are affected by the absorbent, the solvent, the chromatography plate itself, application technique and the temperature of the solvent and plate.What is a mobile and stationary phase?
the mobile phase is the solvent that moves through the paper, carrying different substances with it. the stationary phase is contained on the paper and does not move through it.What solvent is best for paper chromatography?
Readily Available Solvents for Paper Chromatography| Solvent | Polarity (arbitrary scale of 1-5) | Suitability |
|---|---|---|
| Water | 1 – Most polar | Good |
| Rubbing alcohol (ethyl type) or denatured alcohol | 2 – High polarity | Good |
| Rubbing alcohol (isopropyl type) | 3 – Medium polarity | Good |
| Vinegar | 3 – Medium polarity | Good |