.
Thereof, what is the formal charge on the nitrogen in the Lewis structure of no3?
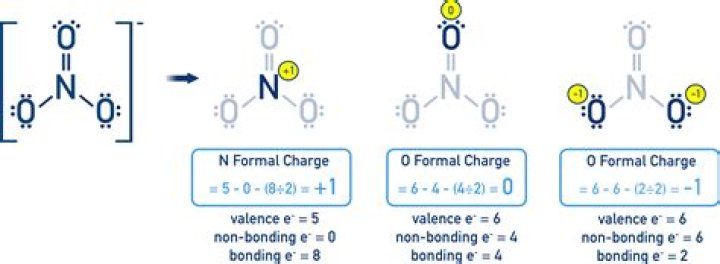
2) NO3– There are 3 possible resonance structures that satisfy the octet rule. In each resonance structure, the formal charge on N = +1; the formal charge on the N = O oxygen = 0; and the formal charge on each of the N – O oxygens = –1.
Furthermore, what is the formal charge on nitrogen in the following structure? The valence electrons of nitrogen in its compounds are all sp³ hybridized orbitals. The formal charge on N is usually -1 for an anion, 0 for a neutral compound, and +1 in cations. A nitrogen atom with a formal charge of -3 would correspond to a nitride ion, N³?, which is strongly basic in aqueous solution.
Beside this, what is the formal charge on nitrogen in the nitrate ion?
For N, we are counting 4 electrons (4 bonding electrons, no nonbonding electrons). Since N is in Group VA, we have one less electron than N should have, and so it has a +1 charge. As a check, your formal charges should add up to the charge of the atom (-1 for nitrate).
What is the formal charge on the nitrogen atom in the nitrate anion no3 -)?
The nitrate ion is a polyatomic anion with the empirical formula NO3- and a molecular mass of 62. 01 daltons; it consists of one central nitrogen atom surrounded by three identical oxygen atoms in a trigonal planar arrangement. The nitrate ion carries a negative one formal charge.
Related Question AnswersWhy is nitrate negatively charged?
Because “nitrate” has a formula of NO3-, its the oxygens that provide the negative charge. The nitrogen atom by itself is normally written as negative, as it needs 3 electrons to be stable. See pictures. Oxygen has 6 electrons and wants 8, so it forms two bonds or one bond and a negative charge.What is co32?
CO3 2- is carbonate. a carbonate is a salt of carbonic acid (H2CO3),characterized by the presence of the carbonate ion, a polyatomic ion with the formula of CO3 2-. CO32- is an anion (a negative ion) seen frequently in chemistry. In the CO32- Lewis structure carbon is the least electronnegative element.What is co3 charge?
With the third oxygen atom , carbon again forms a single bond. As a result , 3rd oxygen atom have 7 electrons and carbon atom have 7+1=8 electrons. Now 2nd and 3rd oxygen atoms have 7–7 electrons . They both needs 2 more electrons. That's why CO3 have a -2 charge.What is the charge of nitrate?
The Formal Charge Of NO3- (Nitrate) Nitrate, chemical formula NO3, has a chemical charge of -1. Ion nitrates have a negative one formal charge. You may be wondering why this is the case.What is the charge of aluminum?
The charge of an aluminum ion is typically 3+. This is because the element's atomic number is 13, reflecting the fact that it has 13 electrons and 13 protons. The valence shell of aluminum has three electrons, and per the octet rule, these three electrons are lost resulting in just 10 electrons and 13 protons.What is the Lewis structure of no3?
For the NO3- Lewis structure, calculate the total number of valence electrons for the NO3- molecule. After determining how many valence electrons there are in NO2, place them around the central atom to complete the octets. In the Lewis structure of NO3- there are a total of 24 valence electrons.What is the hybridization of no3 minus?
Hybridization of NO3 (Nitrate) The hybridization of NO3– is sp2 type. Students will learn about how this hybridization occurs and all the steps involved in it. They will also learn about the molecular geometry and the bond angles of nitrate.Why is no3 nonpolar?
Explanation: I assume you mean NO3- (the nitrate anion); there is no NO3 molecule. It is non-polar because it has a trigonal planar structure and the symmetry means that there is an even distribution of electron charge density over the three N - O bonds.What is the charge of lead?
Group IV A (14) metals form cations with +4 charge, although tin (Sn) and lead (Pb) can form cations having +2 charge.Why is formal charge important?
Knowing the formal charge on a particular atom in a structure is an important part of keeping track of the electrons and is important for establishing and predicting the reactivity. The formal charge on an atom in a molecule reflects the electron count associated with the atom compared to the isolated neutral atom.What does formal charge mean?
In chemistry, a formal charge (FC) is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity.Is hydrogen positive or negative?
One Hydrogen (H) has a positive charge +1. This has nothing to do with it having more protons than electrons, as it has one of each. Think of it as 'willing' to donate one electron, or it has +1 electrons see, it is on the plus side. One Oxygen (O) has a negative charge of -2.How many neutrons are in nitrogen?
7 neutronsIs oxygen positive or negative?
A good example is an oxygen atom that is sharing one electron with each of two nearby hydrogen atoms, making a molecule of water H2O. An oxygen atom that has successfully donated one or two electrons or is sharing one or two electrons has lost part of its negative charge, so it is left positively charged.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.