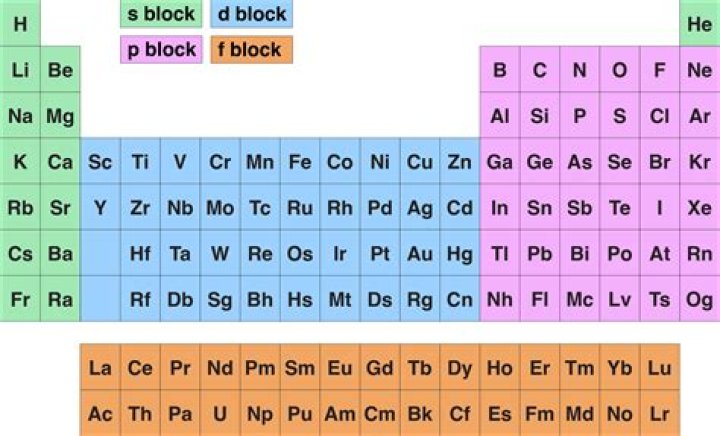
A g block element is a hypothetical element whose ground state electron configuration contains one or more electrons in a g sublevel. Currently there are four blocks on the periodic table: s, p, d & f: These blocks represent the orbital of the last electron listed in the element's electron configuration..
Furthermore, what is a block on the periodic table?
Blocks. A block of the periodic table of elements is a set of adjacent groups. The respective highest-energy electrons in each element in a block belong to the same atomic orbital type. Each block is named after its characteristic orbital: s, p, d, f, g (no elements belonging to the g-block have been observed).
can there be an element 119? Ununennium, also known as eka-francium or element 119, is the hypothetical chemical element with symbol Uue and atomic number 119. In the periodic table of the elements, it is expected to be an s-block element, an alkali metal, and the first element in the eighth period.
Secondly, why is the periodic table divided into blocks?
The elements in this block are all metals, and they react when water is present. The Brainliest Answer! Elements in the long form of periodic table have been divided into four blocks i.e. s ,p ,d and f. This division is based upon the name of the orbitals which receives the last electron.
What is period 4 on the periodic table?
The period 4 transition metals are scandium (Sc), titanium (Ti), vanadium (V), chromium (Cr), manganese (Mn), iron (Fe), cobalt (Co), nickel (Ni), copper (Cu), and zinc (Zn).
Related Question Answers
What does D block mean?
d-block. to steal. I'm going to d-block that kid's sneakers. See more words with the same meaning: to steal.Why is it called the S block?
The s-block elements share electron configurations. s-block elements are the elements found in Group 1 and Group 2 on the periodic table. Because they have 2 valence electrons they are less reactive than group 1. Hydrogen is a nonmetal grouped with the alkali metals because it has one electron in its valence shell.What elements are in the S block?
The s-block elements include hydrogen (H), helium (He), lithium (Li), beryllium (Be), sodium (Na), magnesium (Mg), potassium (K), calcium (Ca), rubidium (Rb), strontium (Sr), cesium (Cs), barium (Ba), francium (Fr) and radium (Ra). The periodic table shows exactly where these elements are within the s-block.What is the big block in the middle of the periodic table called?
The noble gases are called noble because they seldom react with anything. The transition metals make up the middle block of the periodic table. Like the other elements that are grouped together (alkali metals, noble gases), these metals have very similar properties.What are P block elements?
The p-block elements are found on the right side of the periodic table. They include the boron, carbon, nitrogen, oxygen and flourine families in addition to the noble gases. The noble gases have full p-orbital's and are nonreactive. p block orbital.Who discovered halogens?
Scheele called the element "dephlogisticated muriatic acid", which is how chlorine was known for 33 years. In 1807, Humphry Davy investigated chlorine and discovered that it is an actual element.What are the S and P blocks called?
The elements of “s” and “p” blocks except “d” group elements are called as representative elements because Their outer shells are not completely filled with electrons and The elements get the nearest inert gas configuration by losing or gaining or sharing of electrons. They are chemically active.What is the point of electron configuration?
Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating with the loss of or gain of electrons in their subsequent orbitals.Why does Period 4 have 18 elements?
The third period is similar to the second, filling the 3s and 3p sublevels. Notice that the 3 d sublevel does not actually fill until after the 4s sublevel. This results in the fourth period containing 18 elements due to the additional 10 electrons that are contributed by the d sublevel.How many block are there in periodic table?
four blocks
What groups make up the D block?
d-Block Elements. The d-block elements are found in groups 3, 4, 5, 6, 7, 8, 9, 10, 11, and 12 of the periodic table; d-block elements are also known as the transition metals. The d orbital is filled with the electronic shell “n-1.”Why are orbitals called SPDF?
The orbital names s, p, d, and f stand for names given to groups of lines originally noted in the spectra of the alkali metals. These line groups are called sharp, principal, diffuse, and fundamental.What is the number of valence electrons in phosphorus?
five
How many elements are there?
118 elements
What is a period number on the periodic table?
Periods in the periodic table. In each period (horizontal row), the atomic numbers increase from left to right. The periods are numbered 1 through 7 on the left-hand side of the table. Elements that are in the same period have chemical properties that are not all that similar.How does the Aufbau principle work?
The Aufbau principle, simply put, means electrons are added to orbitals as protons are added to an atom. The term comes from the German word "aufbau", which means "built up" or "construction". Lower electron orbitals fill before higher orbitals do, "building up" the electron shell.What is SPDF chemistry?
s, p, d, f and so on are the names given to the orbitals that hold the electrons in atoms. These orbitals have different shapes (e.g. electron density distributions in space) and energies (e.g. a hydrogen atom with one electron would be denoted as 1s1 - it has one electron in its 1s orbital.What is element 140?
Unquadnilium, also known as eka-thorium or element 140, is the temporary name of a hypothetical superheavy chemical element in the periodic table in the superactinide series that has the temporary symbol Uqn and has the atomic number 140. In the periodic table of elements, it is a f-block element.Is there an element 123?
Murinite (symbol Mu) is a chemical element, atomic number 123 on the periodic table.