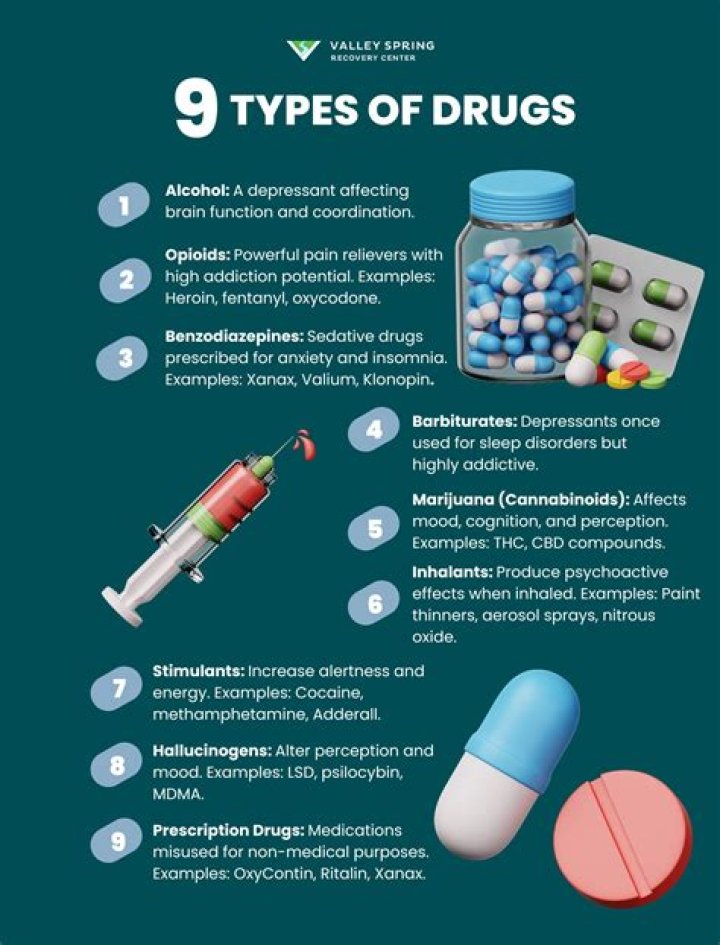
Acetic acid /?ˈsiːt?k/, systematically named ethanoic acid /ˌ?θ?ˈno??k/, is a colourless liquid organic compound with the chemical formula CH3COOH (also written as CH3CO2H or C2H4O2). When undiluted, it is sometimes called glacial acetic acid..
Beside this, what is the purpose of acetic acid?
Acetic acid has many functions, but it is mostly used as a chemical reagent, fungicide, herbicide, microbiocide, pH adjuster, counterirritant, and solvent in a variety of industries, including, but not limited to food, agriculture, cleaning, and cosmetics.
Likewise, is acetic acid harmful to humans? Acetic Acid Hazards. Acetic acid can be a hazardous chemical if not used in a safe and appropriate manner. This liquid is highly corrosive to the skin and eyes and, because of this, must be handled with extreme care. Acetic acid can also be damaging to the internal organs if ingested or in the case of vapor inhalation.
Likewise, what is acetic acid in?
Acetic acid is also known as the second simplest carboxylic acid. Acetic acid is most popularly known because of its use in vinegar. A majority of the acetic acid produced is used to produce vinyl acetate monomer (VAM), which is the building block to make paints, adhesives, packaging and more.
What foods contain acetic acid?
It has found use in a variety of products, including condiments such as ketchup, mustard, mayonnaise, and relish, salad dressings, marinades for meat, poultry, and fish, bakery products, soups, and cheeses.
Related Question Answers
Is acetic acid strong or weak?
Acetic acid is a weak acid because it is not a strong acid which has a specific definition in chemistry: Strong acids completely dissociate in aqeous solution, that is, all their H+ come off in water. H+ is also called a proton because hydrogen without an electron is essentially a proton.Where is acetic acid found naturally?
Ingredient Results - Acetic Acid. A naturally occurring acid found in a variety of plants and fruits such as apples, grapes, oranges, pineapples, and strawberries. It is an organic acid that gives vinegar its sour taste and distinct smell. It Is produced through a fermentation process.Is Vinegar an oxidizer?
Vinegar is an acid (in fact, it's known as acetic acid). The hypochlorous acid then reacts with the rest of the vinegar, Dr. Biberdorf continues. Because hypochlorous acid is something that's called an oxidizing agent, it takes electrons from whatever it comes into contact with.What is the natural source of acetic acid?
Name the natural source of Acetic acid. Oxalic acid is found in tomatoes, lactic acid in curd and acetic acid is found in vinegar.What can acetic acid dissolve?
Liquid acetic acid is a hydrophilic ( polar) protic solvent, similar to ethanol and water. With a moderate dielectric constant of 6.2, it can dissolve not only polar compounds such as inorganic salts and sugars, but also non-polar compounds such as oils and elements such as sulfur and iodine.Is acetic acid flammable?
Acetic acid is a colourless liquid; with a strong vinegar-like odour. It is flammable, and at temperatures warmer than 39°C, explosive vapour/air mixtures may be formed.What is the pH of acetic acid?
Acetic acid is a weak monoprotic acid. In aqueous solution, it has a pKa value of 4.76. Its conjugate base is acetate (CH3COO−). A 1.0 M solution (about the concentration of domestic vinegar) has a pH of 2.4, indicating that merely 0.4% of the acetic acid molecules are dissociated.Why is acetic acid good for cleaning?
Vinegar's acidity is what makes it such a good cleaner. Because vinegar is so acidic, it can counteract some icky buildups. It can dissolve away soap scum, brines left by hard water, and glue left behind by stickers. A paste of vinegar and baking soda can dissolve set red wine stains.How Acetic acid is manufactured?
Industrially, acetic acid is produced both synthetically and by bacterial fermentation. Approximately 75% of acetic acid used in the chemical industry is made by the carbonylation of methanol. Most acetic acid is made by methanol carbonylation, where methanol and carbon monoxide react to produce acetic acid.Is COOH an acid or base?
Carboxyl groups are weak acids, dissociating partially to release hydrogen ions. The carboxyl group (symbolized as COOH) has both a carbonyl and a hydroxyl group attached to the same carbon atom, resulting in new properties.Can acetic acid be stored in plastic?
I have stored glacial acetic acid in PET plastic for years with absolutely no problems. With PET plastic you should not store liquids that are intensely alkaline (like household bleach), as it softens the plastic to the point of breakage.Who discovered acetic acid?
The first person to extract acetic acid from vinegar was the Muslim alchemist Jabir ibn Hayyan Geber (c. 721–815). The pure compound was not produced, however, for another ten centuries when the German chemist Georg Ernst Stahl (1660–1734) extracted acetic acid from vinegar in 1700 by distillation.What is acetic acid used for medically?
Acetic acid (medical use) Acetic acid, which at low concentrations is known as vinegar, is an acid used to treat a number of conditions. As an eardrop it is used to treat infections of the ear canal. It may be used with an ear wick.How is acetic acid useful in the home?
Household Uses Acetic acid which is a dilute solution is used extensively as vinegar. And as we are familiar, vinegar is widely used for cleaning, laundry, cooking, and many other household uses. Farmers usually spray acetic acid on livestock silage to counter bacterial and fungal growth.How glacial acetic acid is prepared?
Removing the water from acetic acid lowers its melting point by 0.2 °C. Glacial acetic acid may be prepared by dripping acetic acid solution over a "stalactite" of solid acetic acid (which could be considered to be frozen).What is the Colour of acetic acid?
colourless
Why does acetic acid have an odor?
Acetic acid. Acetic acid is an organic compound formed in lambic by the oxidation of ethanol. An organic acid, it is a colorless liquid in its pure state with a strong acidic odor. In more dilute solution, the odor can take on a smell of overripe fruit.What is the chemical formula of vinegar?
CH3COOH
Is vinegar acetic acid?
Vinegar is an aqueous solution of acetic acid and trace chemicals that may include flavorings. Usually the acetic acid is produced by the fermentation of ethanol or sugars by acetic acid bacteria.